Introduction
It is now generally accepted that sickle cell trait is protective against malaria. People with sickle cell anemia (SCA) also have been shown to have a lower incidence of malaria compared to the general population, though the combination of SCA and malaria significantly increases risk of severe malaria, thereby posing a higher risk for mortality [1]. Malaria worsens the anemia associated with SCA, potentially causing life-threatening complications [2,3]. Additionally, the invasion of red blood cells (RBCs) by malaria parasites triggers extensive sickling and increases the adhesion of infected sickle cells to blood vessel walls, promoting vaso-occlusive crises (VOCs) [4]. Severe VOC can subsequently lead to acute chest syndrome (ACS), characterized by fever, respiratory distress, and new lung infiltrates on chest X-rays. We report a case of severe Plasmodium falciparum infection in a patient with SCA complicated by possible ACS, which was successfully treated with intravenous (IV) artesunate and RBC exchange transfusion.
Case Report
A 48-year-old male with a past medical history of SCA and pulmonary embolism presented with fever and chills for 4 days. He endorsed shortness of breath, diffuse body aches, worse in the legs, as well as nausea and diarrhea. He had returned from a trip to Nigeria 2 to 3 weeks earlier and denied taking prophylactic treatment for malaria. Folic acid was the only home medication he was taking.
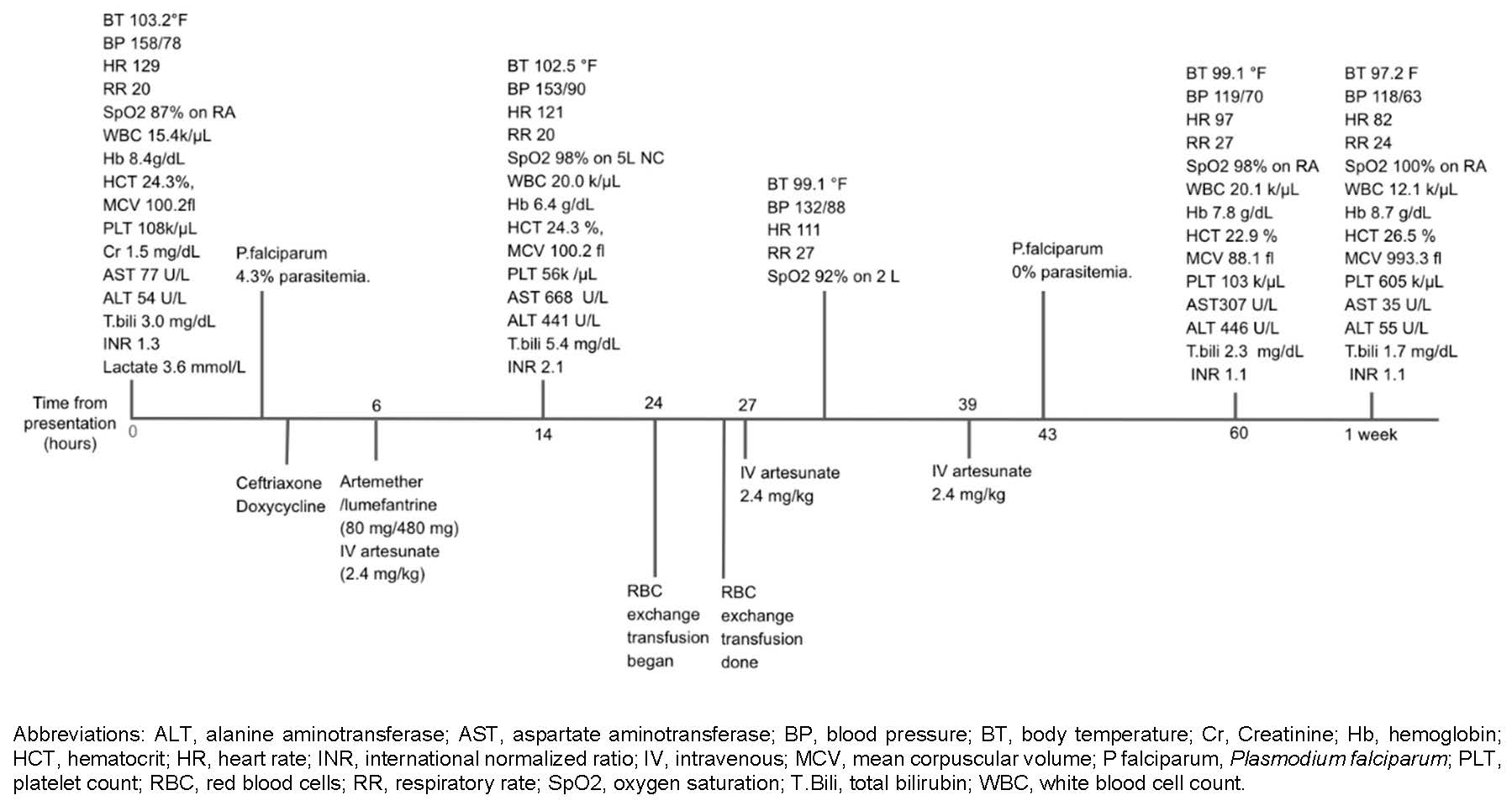
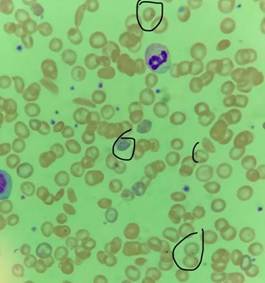
Initial vital signs showed tachycardia at 129, normal blood pressure, fever at 103.2 °F, and oxygen saturation of 87% on room air. On physical exam, scleral icterus and epigastric tenderness were noted. Initial labs were significant for leukocytosis (white blood cells, 15.4 k/μL), macrocytic anemia (hemoglobin [Hb], 8.4 g/dL; hematocrit, 24.3%; mean corpuscular volume, 100.2 fl), thrombocytopenia (platelet count, 108 k/μL), elevated creatinine (1.5 mg/dL), mild transaminitis (aspartate aminotransferase [AST], 77 U/L; alanine aminotransferase [ALT], 54 U/L), hyperbilirubinemia (total bilirubin 3.0 mg/dL), elevated international normalized ratio (INR), 1.3; and elevated lactate (3.6 mmol/L) (Figure 1). Chest X-ray showed mild streaky perihilar predominant bilateral opacities suspicious for edema. Blood parasite thick and thin smears showed P falciparum with 4.3% parasitemia (Figure 2). The patient was diagnosed with severe malaria given pulmonary edema, lactic acidosis, acute kidney injury, and elevated parasitemia.


Treatment was initiated first with artemether/lumefantrine (80 mg/480 mg), and then IV artesunate (2.4 mg/kg) once it was obtained by the hospital. Given increasing oxygen requirement to 5 L through nasal cannula, tachypnea to respiratory rate of 20 and new chest X-ray finding with bilateral mild interstitial edema, concern was also raised for ACS, so empiric therapy for community-acquired pneumonia with ceftriaxone and doxycycline were promptly started as well.
The next morning, the patient remained febrile with worsening transaminitis (AST, 668 U/L; ALT, 441 U/L) and hyperbilirubinemia (total bilirubin, 5.4 mg/dL), increasing INR to 2.1, and worsening Hb to 6.4g/dL and thrombocytopenia to 56 k/μL. His hypoxia also did not improve and he appeared more drowsy. Due to concern for worsening ACS, hematology recommended proceeding with RBC exchange transfusion. The patients second dose of artesunate was due near the end of RBC exchange so it was given after the exchange completed due to concern about possible clearance of artesunate if given during the exchange. A repeat blood smear taken 4 hours after the third dose showed parasitemia reduced to 0%.The patients fever began to subside after the second dose of artesunate and exchange transfusion. Follow-up laboratory tests revealed stabilized liver function, and improved anemia and thrombocytopenia. Treatment was then transitioned to artemether-lumefantrine for an additional 3-day course (Figure 1).
Discussion
According to the Centers for Disease Control and Prevention (CDC), severe malaria is defined by the presence of one or more of the following: high parasitemia (≥5%), severe anemia (Hb < 7 g/dL), impaired consciousness, seizures, shock, pulmonary edema, acute respiratory distress syndrome, acidosis, acute kidney injury, abnormal bleeding, disseminated intravascular coagulation, or jaundice if accompanied by other symptoms [5]. Severe malaria has a mortality rate exceeding 5%, increasing the risk of death by over fifty times [6].
Sickle cell disease (SCD), an autosomal recessive disorder characterized by chronic hemolytic anemia and painful VOCs, makes patients more susceptible to severe infections, including malaria [2,7]. In patients with SCDs, malaria not only exacerbates preexisting anemia to life-threatening levels but also impairs splenic function, hindering the clearance of parasitized red blood cells [8]. Research has shown that patients with sickle cell disease experience higher rates of severe malaria-induced anemia [3,9,10] and that malaria significantly contributes to early mortality in these individuals. Additionally, infections like malaria can trigger ACS, a severe complication caused by vaso-occlusion within the pulmonary vasculature, which can be life-threatening [11].
To prevent fatal outcomes in patients presenting with severe malaria, the CDC emphasizes the urgent need for immediate IV antimalarial therapy [5]. Artesunate is the only available IV option in the United States, and its administration should begin as soon as possible [12]. If IV artesunate is not immediately accessible, oral antimalarials such as artemether-lumefantrine may be used temporarily until the IV medication is obtained. Artesunate for Injection™ can be acquired through the manufacturer, Amivas, which offers 24/7 support to help hospital pharmacies locate the nearest distributor.
The mechanism of action of IV artesunate is to convert to dihydroartemisinin (DHA), which targets red blood cells by inhibiting heme polymerization, generating reactive oxygen species, destabilizing parasite membranes, and damaging parasite proteins, leading to parasite death [13]. IV artesunate is administered at a dose of 2.4 mg/kg at 0, 12, and 24 hours. After completing this initial course, a thin blood smear is performed 4 hours after the last dose to determine parasite density. If the density is ≤1% and the patient can tolerate oral medications, follow-up treatment with oral artemether-lumefantrine should be initiated within 4 to 24 hours [14]. If the parasite density remains >1% after the third dose, IV artesunate must be continued once daily for up to 7 days or until the density decreases to ≤1%. Once the parasite density is reduced to ≤1% and the patient is stable enough for oral medications, a complete follow-up oral regimen should be administered to ensure eradication of the parasite.
Although rare, delayed hemolytic anemia has been reported in some patients following IV artesunate therapy, particularly in patients with high parasite densities. To mitigate this potential complication, weekly monitoring for signs of hemolytic anemia is recommended for up to 4 weeks after the initiation of treatment [15,16]. In cases such as this one, where the disease course was further complicated by worsened anemia due to the patients underlying SCD, close monitoring for delayed hemolytic anemia was particularly crucial.
Severe P falciparum malaria continues to have a high case fatality rate despite the availability of effective antimalarial and supportive therapies. Approaches like removing infected red blood cells and plasma with exchange transfusion have been explored as potential treatments for severe cases [17,18]. Although exchange transfusion has shown success in individual instances, its efficacy remains debated. Evidence from retrospective studies and meta-analyses suggests that exchange transfusion does not improve survival outcomes in severe malaria, and therefore, it is not recommended as a treatment option [19–22].
In our case, exchange transfusion was performed as recommended for ACS under specific conditions, including a history of severe ACS or cardiopulmonary disease, multilobar involvement, rapidly progressing ACS or multiorgan failure, and severe hypoxemia (SaO2 ≤ 85% or PaO2 ≤ 55 mm Hg) [19,23–25]. There are no established guidelines on the dosing and timing of IV artesunate in cases requiring exchange transfusion, as these interventions may need to be administered simultaneously in patients with severe VOC from SCD and severe malaria.
In the body, both artesunate and its metabolite, DHA, are approximately 93% protein-bound in plasma, with IV artesunate having a short half-life of under 15 minutes and DHA being eliminated within 30 to 60 minutes [26]. Given that artesunate is primarily protein-bound in plasma, the reinfusion of the plasma component during exchange transfusion should lead to minimal loss of the drug. However, a theoretical risk of artesunate removal remains as it distributes into RBCs. If possible, to decrease this risk, we suggest administering the drug prior to initiating the exchange procedure, with a waiting period of at least 1 to 2 hours before starting the transfusion. Further studies are warranted to better understand the interaction between exchange transfusion procedures and the pharmacokinetics of IV artesunate.
Conclusion
Our study highlights a case of severe P falciparum malaria complicated by ACS in a patient with underlying SCA, successfully managed with simultaneous exchange transfusion and 3 doses of IV artesunate. This case underscores the potential need for concurrent administration of exchange transfusion and IV artesunate, as SCD predisposes patients to severe malaria, while severe malaria, in turn, increases the risk of VOC requiring exchange transfusion.
Careful consideration must be given to the timing and administration of IV artesunate during exchange transfusion due to the theoretical risk of medication loss during the procedure. Further research is necessary to better understand the interaction between exchange transfusion and the pharmacokinetics of IV artesunate to optimize treatment strategies.
Author Contributions
Conceptualization, L.K. and L.H.; methodology, L.K. and L.H.; Resources, L.K., L.S., and L.H.; Data curation, L.K. and L.S.; writing—original draft preparation, L.K.; writing—review and editing, L.K., L.S., and L.H.; supervision, L.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Uyoga, S.; Olupot-Olupot, P.; Connon, R.; Opoka, R.O.; Alaroker, F.; Muhindo, R.; Macharia, A.W.; Dondorp, A.M.; Gibb, D.M.; et al. Sickle cell anaemia and severe Plasmodium falciparum malaria: A secondary analysis of the Transfusion and Treatment of African Children Trial (TRACT). Lancet Child Adolesc. Health 2022, 6, 606–613. [CrossRef] [PubMed]
- Makani, J.; Komba, A.N.; Cox, S.E.; Oruo, J.; Mwamtemi, K.; Kitundu, J.; Magesa, P.; Rwezaula, S.; Meda, E.; Mgaya, J.; et al. Malaria in patients with sickle cell anemia: Burden, risk factors, and outcome at the outpatient clinic and during hospitalization. Blood 2010, 115, 215–220. [CrossRef] [PubMed]
- Eleonore, N.L.E.; Cumber, S.N.; Charlotte, E.E.; Lucas, E.E.; Edgar, M.M.L.; Nkfusai, C.N.; Geh, M.M.; Ngenge, B.M.; Bede, F.; Fomukong, N.H.; et al. Malaria in patients with sickle cell anaemia: Burden, risk factors and outcome at the Laquintinie hospital, Cameroon. BMC Infect. Dis. 2020, 20, 40. [CrossRef] [PubMed]
- Ahmed, S.G. The role of infection in the pathogenesis of vaso-occlusive crisis in patients with sickle cell disease. Mediterr. J. Hematol. Infect. Dis. 2011, 3, e2011028. [CrossRef]
- Clinical Features of Malaria. US Centers for Disease Control and Prevention. 2024. Available online: https://www.cdc.gov/malaria/hcp/clinical-features/index.html (accessed on 31 December 2024).
- White, N.J. Severe malaria. Malar. J. 2022, 21, 284. [CrossRef]
- McAuley, C.F.; Webb, C.; Makani, J.; Macharia, A.; Uyoga, S.; Opi, D.H.; Ndila, C.; Ngatia, A.; Scott, J.A.G.; Marsh, K.; et al. High mortality from Plasmodium falciparum malaria in children living with sickle cell anemia on the coast of Kenya. Blood 2010, 116, 1663–1668. [CrossRef]
- Luzzatto, L. Sickle cell anaemia and malaria. Mediterr. J. Hematol. Infect. Dis. 2012, 4, e2012065. [CrossRef]
- Ngolet, L.O.; Moyen Engoba, M.; Kocko, I.; Elira Dokekias, A.; Mombouli, J.V.; Moyen, G.M. Sickle-cell disease healthcare cost in Africa: Experience of the Congo. Anemia 2016, 45, 2046535. [CrossRef]
- Aninagyei, E.; Doku Tetteh, E.; Banini, J.; Nani, E.; Adu, P.; Ephraim, R.K.D.; Egyir-Yawson, A.; Acheampong, D.O. Haematological parameters and plasma levels of 8-iso-prostaglandin F2α in malaria-sickle cell co-morbidity: A cross sectional study. bioRxiv 2018. [CrossRef]
- Bhasin, N.; Sarode, R. Acute chest syndrome in sickle cell disease. Transfus. Med. Rev. 2023, 37, 150755. [CrossRef]
- Dondorp, A.M.; Fanello, C.I.; Hendriksen, I.C.; Gomes, E.; Seni, A.; Chhaganlal, K.D.; Bojang, K.; Olaosebikan, R.; Anunobi, N.; Maitland, K.; et al. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): An open-label, randomised trial. Lancet 2010, 376, 1647–1657. [CrossRef]
- Ruwizhi, N.; Maseko, R.B.; Aderibigbe, B.A. Recent advances in the therapeutic efficacy of artesunate. Pharmaceutics. 2022, 14, 504. Ruwizhi N, Maseko RB, Aderibigbe BA. Recent advances in the therapeutic efficacy of artesunate. Pharmaceutics 2022, 14, 504. [CrossRef]
- Griffith, K.S.; Lewis, L.S.; Mali, S.; Parise, M.E. Treatment of malaria in the United States: A systematic review. JAMA 2007, 297, 2264–2277. [CrossRef]
- Gómez-Junyent, J.; Ruiz-Panales, P.; Calvo-Cano, A.; Gascón, J.; Muñoz, J. Delayed haemolysis after artesunate therapy in a cohort of patients with severe imported malaria due to Plasmodium falciparum. Enferm. Infecc. Microbiol. Clín. 2017, 35, 516–519. [CrossRef]
- Jauréguiberry, S.; Ndour, P.A.; Roussel, C.; Ader, F.; Safeukui, I.; Nguyen, M.; Biligui, S.; Ciceron, L.; Mouri, O.; Kendjo, E.; et al. Postartesunate delayed hemolysis is a predictable event related to the lifesaving effect of artemisinins. Blood 2014, 124, 167–175. [CrossRef] [PubMed]
- Zhang, J.; Chen, L.; Zhang, M.; Yao, M.; Ren, S.; Liu, H.; Min, Y.; Jia, Y.; Tao, Y.; Zhang, H. Exchange transfusion combined with artesunate (ET-AS) as a safe and effective therapy in severe P. falciparum malaria: A case series. BMC Infect. Dis. 2024, 24, 603. [CrossRef]
- Dongare, H.C.; Khatib, K.I. Exchange transfusion in severe falciparum malaria. J. Clin. Diagn. Res. 2016, 10, OD05–OD06. [CrossRef]
- Schwartz, J.; Winters, J.L.; Padmanabhan, A.; Balogun, R.A.; Delaney, M.; Linenberger, M.L.; Szczepiorkowski, Z.M.; Williams, M.E.; Wu, Y.; Shaz, B.H. Guidelines on the use of therapeutic apheresis in clinical practice—Evidence-based approach from the writing committee of the American Society for Apheresis: The sixth special issue. J. Clin. Apher. 2013, 28, 145–284. [CrossRef]
- Kreeftmeijer-Vegter, A.R.; Melo, M.D.M.; De Vries, P.J.; Koelewijn, R.; Van Hellemond, J.J.; Van Genderen, P.J. Manual blood exchange transfusion does not significantly contribute to parasite clearance in artesunate-treated individuals with imported severe Plasmodium falciparum malaria. Malar. J. 2013, 12, 115. [CrossRef]
- Panosian, C.B. Editorial Response: Exchange blood transfusion in severe falciparum malaria—The debate goes on. Clin. Infect. Dis. 1998, 26, 853–854. [CrossRef]
- Riddle, M.S.; Jackson, J.L.; Sanders, J.W.; Blazes, D.L. Exchange transfusion as an adjunct therapy in severe Plasmodium falciparum malaria: A meta-analysis. Clin. Infect. Dis. 2002, 34, 1192–1198. [CrossRef] [PubMed]
- Mwaiswelo, R.O.; Mawala, W.; Iversen, P.O.; De Montalembert, M.; Luzzatto, L.; Makani, J. Sickle cell disease and malaria: Decreased exposure and asplenia can modulate the risk from Plasmodium falciparum. Malar. J. 2020, 19, 165. [CrossRef] [PubMed]
- Paul, R.N.; Castro, O.L.; Aggarwal, A.; Oneal, P.A. Acute chest syndrome: Sickle cell disease. Eur. J. Haematol. 2011, 87, 191–207. [CrossRef] [PubMed]
- Mamdapur, A.B.; Sagar, M.S.; Madhusudan, R.; Samir, M. Therapeutic red cell exchange transfusion as an adjuvant therapy for management of sickle cell crisis in adults. Indian J. Crit. Care Med. 2018, 22, 457–459. [CrossRef]
- Morris, C.A.; Duparc, S.; Borghini-Fuhrer, I.; Jung, D.; Shin, C.S.; Fleckenstein, L. Review of the clinical pharmacokinetics of artesunate and its active metabolite dihydroartemisinin following intravenous, intramuscular, oral or rectal administration. Malar. J. 2011, 10, 263. [CrossRef]

