Introduction
Disseminated histoplasmosis is a severe manifestation of infection by the fungus Histoplasma capsulatum, characterized by its spread from the lungs to multiple organs, often in immunocompromised individuals [1]. Historically, the disease has been associated with outbreaks in endemic areas, where exposure to contaminated soil or bird droppings leads to inhalation of fungal spores. Once disseminated, histoplasmosis presents with nonspecific symptoms such as fever, weight loss, hepatosplenomegaly, and bone marrow suppression, making early diagnosis challenging [2]. Diagnosing disseminated histoplasmosis requires a high index of suspicion, a compatible clinical picture, and knowledge of appropriate diagnostic tests. Antigen detection and molecular techniques have improved the speed and accuracy of diagnosis in recent years. Although standard diagnostic methods have evolved, fungal cultures and serologic testing remain central to diagnosis confirmation [3,4].
This case highlights the diagnostic challenges posed by disseminated histoplasmosis, particularly in patients with atypical presentations or no underlying immunosuppression. Clinical suspicion continues to remain crucial in regions where the disease is endemic. The case also emphasizes the importance of antifungal therapy, as well as the intrinsic barriers associated with the long-term management required to treat this disease. Early, targeted therapy is critical because delays can lead to high morbidity and mortality, as illustrated by past outbreaks [5].
Our case adds to the current literature by emphasizing the need for a multidisciplinary diagnostic approach, combining traditional and novel techniques, and the importance of early intervention. As diagnostic modalities advance, particularly with antigen and molecular testing, there is a need to refine treatment protocols to reduce relapse rates and improve long-term outcomes for patients with disseminated histoplasmosis.
Case Presentation
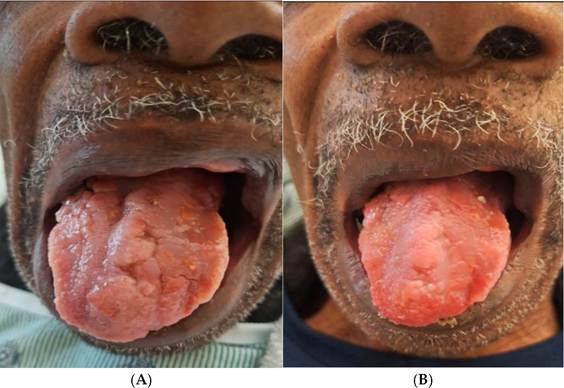
A 73-year-old African American male, originally from Alabama, presented with chronic oral lesions and odynophagia. He was initially diagnosed with herpes simplex virus and was treated with oral valacyclovir and an oral mouthwash in March 2024. However, symptoms never fully resolved, leading to a reduced oral intake, a weight loss of 20 pounds over a few months, and persistent pain.
Upon admission, the patient was afebrile with stable vital signs, including a blood oxygen saturation of 97% on room air. The Infectious Disease (ID) team was consulted due to concerns about a herpes zoster infection. His laboratory tests revealed a white blood cell count of 3.4, with a comprehensive metabolic panel within normal limits.
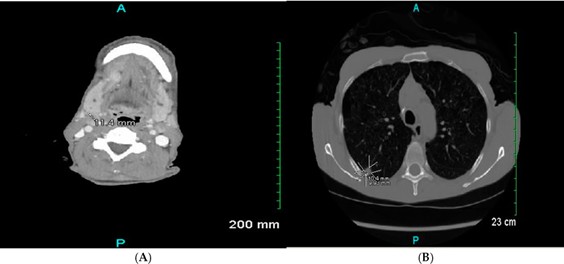
A computed tomography (CT) scan of the neck revealed mildly enlarged and atypical cervical lymph nodes, raising concerns for metastatic disease or an atypical infection, along with a suspicious right upper lobe pulmonary nodule in the setting of moderate centrilobular emphysema. He was also found to have periorbital soft tissue edema and left thyroid goiter. A chest CT identified a right upper lobe nodule with suspicious background emphysematous change [Figure 1].

The patient had a past medical history of prostate cancer and a surgical history of hernia repair. He was not taking any prescription medications, instead opting for over-the-counter drugs. His social history was notable for being a former smoker with no alcohol use. He denied intravenous drug use and recent travel. He lived alone and had no pets; his family history was noncontributory.
A review of systems revealed a significant weight loss of 20 pounds, along with oral lesions and painful swallowing. His physical examination showed oral ulcers and mild oral thrush, while the rest of his systems were unremarkable, including normal lymph nodes and stable cardiovascular, respiratory, and neurological findings.
His initial assessment was not compatible with herpes zoster infection. The Ear, Nose, and Throat specialist performed a laryngoscope and biopsy of the oral lesions. The scope was unremarkable overall. Pathology from the biopsy, however, revealed yeast consistent with a Histoplasma infection.
Upon further inquiry about his occupational history, the patient disclosed he was an arborist for 14 years. This occupation was the likely source of his exposure to the fungus.
A fungal workup revealed positive antibodies for Histoplasma with a titer of 1:16 by complement fixation, along with a borderline positive result for Aspergillus antibodies and a positive Blastomyces/Coccidioides serology. Cryptococcal, Aspergillus, and Histoplasma antigens were all negative. The Fungitell (1-3)-ß-D-glucan assay test was positive at levels of more than 500 pg/mL. HIV testing was negative, and herpes simplex virus type 2 immunoglobulin G was positive.
Further imaging studies were performed, including a CT scan of the chest, abdomen, and pelvis, which revealed a stable 1 cm right upper lobe nodule that continued to suggest the possibility of neoplasm. There were also bilateral indeterminable adrenal masses with enlarged heterogeneous left thyroid and substernal extension that could not rule out metastatic disease. A magnetic resonance imaging of the abdomen confirmed the bilateral adrenal masses. The pulmonary specialist was also consulted for the lung lesion noted, and recommendations for interventional radiology biopsy were given.
An adrenal biopsy showed a non-necrotizing granulomatous inflammation with organisms morphologically consistent with H capsulatum but negative for malignancy. The lung lesion was also suspected to be related to Histoplasma rather than malignancy. The oral biopsy was sent for fungal culture and ultimately was positive for H capsulatum.
The patient was diagnosed with disseminated histoplasmosis, given oral lesions and adrenal lesions. Both biopsies were proven with Histoplasma, as well as presumptive lung lesion with the same, but no pathology was confirmed.
The patient was started on intravenous liposomal amphotericin B at 3 mg/kg daily for 2 weeks, followed by a 12-month course of oral itraconazole [Figure 2].
Discussion
H capsulatum is the most common cause of endemic fungal infections in the United States. While many infections are either asymptomatic or self-limiting, some patients develop acute pulmonary infections or severe disseminated diseases. Histoplasma is a dimorphic fungus, existing as a mold in the environment and transitioning to a yeast form at body temperature (37 ˝C) once inhaled into the hosts lungs. Traditionally, Histoplasma is endemic in the Ohio and Mississippi river valleys, but research has shown its extensive and global nature [6]. The infection begins when Histoplasma microconidia are inhaled, transforming into yeasts that trigger an immune response involving neutrophils, macrophages, lymphocytes, and natural killer cells. Macrophages play a key role in disseminating the fungus through the lymphatic system to adjacent lymph nodes and, subsequently, via the bloodstream to organs of the reticuloendothelial system, such as the liver, spleen, lymph nodes, and bone marrow. In cases of disseminated infection, macrophages become engorged with yeast, although organisms may also be found in tissue spaces and peripheral white blood cells [1].

This case contributes to clinician knowledge regarding histoplasmosis, particularly concerning its presentation in older adults with no overt immunosuppression. Previous research has emphasized the risk factors for severe disease in immunocompromised individuals, such as those undergoing organ transplantation, patients receiving tumor necrosis factor-alpha inhibitors such as infliximab, etanercept, or adalimumab, and individuals with HIV. Other risk factors include primary and acquired immunodeficiency, taking other immunosuppressive medications such as glucocorticoids, and extremes of age. However, this case illustrates that histoplasmosis can present as a chronic, disseminated infection in middle-aged and older men without known immunosuppression, likely due to unidentified defects in cellular immunity. This broadens the clinical understanding of the population at risk and aligns with findings in older case studies [1].
The clinical manifestations of histoplasmosis are variable, largely determined by the patients immune status and the degree of exposure. Some patients present soon after exposure, while others may develop symptoms years later. Disseminated histoplasmosis presents differently depending on the host: severely immunocompromised patients may experience shock, acute renal failure, hepatic and renal failure, altered mental status, and coagulopathy, while those with chronic infection may exhibit pancytopenia, hepatosplenomegaly, elevated liver function tests, and oropharyngeal lesions. Other sites of infection include the skin, brain, and adrenal glands. This case highlights the importance of recognizing both the acute and chronic forms of disseminated histoplasmosis, especially in older adults who may not exhibit the classic risk factors for severe disease [2].
Diagnosing disseminated histoplasmosis is complex and relies on identifying risk factors, recognizing clinical manifestations, and confirming the presence of Histoplasma through laboratory testing. Diagnosis can be supported by histopathological examination, cultures, antigen detection, and Histoplasma-specific antibody tests [3,4]. Antigen detection tests for H capsulatum have a sensitivity of 60% to 83% of patients with acute pulmonary histoplasmosis [7]. The highest sensitivity is obtained by testing both urine and serum [8]. Cross-reactions are often seen in patients with blastomycosis [7]. Serology testing can be performed using complement fixation, immunodiffusion, and enzyme immunoassay. Complement fixation is slightly more sensitive, but less specific, than the immunodiffusion test (sensitivity 95% vs 90%) [9].
In this case, the patients occupational exposure and age were likely significant risk factors, emphasizing the importance of a thorough occupational history in assessing potential exposures.
In terms of treatment, disseminated histoplasmosis is managed according to the severity of the disease. The Infectious Diseases Society of America (IDSA) last updated clinical guidelines for the management of histoplasmosis in 2007 [10]. Moderate to severe cases are treated with liposomal amphotericin B for 1 to 2 weeks, followed by oral itraconazole for at least 12 months. Mild to moderate cases are treated only with oral itraconazole for the same duration. For central nervous system involvement, liposomal amphotericin B is administered for 4 to 6 weeks, followed by oral itraconazole for at least 12 months, with close monitoring of Histoplasma antigen levels in blood or cerebrospinal fluid. The current case mirrors the literature regarding the recommended treatment protocol [5]. Although the distribution of Histoplasma infections outside typical endemic regions is on the rise, in a recent article, a significant number of ID physicians reported not having seen patients with histoplasmosis, though the ones that had experience with this infection reported adherence to IDSA guidelines, thus showing the crucial need for new histoplasmosis guidelines generally, and for immunocompromised populations in particular [11].
Our patient was lost to follow-up, adding another difficult dimension to the management challenges of histoplasmosis. Our patient did not return for his scheduled monitoring of itraconazole levels. This highlights the critical importance of ongoing surveillance in managing histoplasmosis, particularly for long-term therapy. Without adequate follow-up, there is an increased risk of treatment failure, recurrence, or complications associated with disseminated infection.
Conclusion
Our case of disseminated histoplasmosis in an immunocompetent older male emphasizes the importance of considering fungal infections in patients with chronic symptoms and atypical radiographic findings, even in the absence of classic risk factors such as immunosuppression. The patients occupational history and delayed presentation broaden our understanding of populations at risk of severe Histoplasma infections. Future studies should explore potential subclinical immunological impairments in older adults, possibly predisposing them to disseminated disease. Our case also highlights the critical need for strict adherence to long-term antifungal therapy and close monitoring to prevent disease recurrence.
Funding
This research received no external funding.
Conflicts of Interest
The author declares no conflict of interest.
References
- Goodwin, R.A., Jr.; Shapiro, J.L.; Thurman, G.H.; Thurman, S.S.; Des Prez, R.M. Disseminated histoplasmosis: Clinical and pathologic correlations. Medicine 1980, 59, 1–33. [CrossRef] [PubMed]
- Assi, M.A.; Sandid, M.S.; Baddour, L.M.; Roberts, G.D.; Walker, R.C. Systemic histoplasmosis: A 15-year retrospective institutional review of 111 patients. Medicine 2007, 86, 162–169. [CrossRef] [PubMed]
- Sathapatayavongs, B.; Batteiger, B.E.; Wheat, J.; Slama, T.G.; Wass, J.L. Clinical and laboratory features of disseminated histoplasmosis during two large urban outbreaks. Medicine 1983, 62, 263–270. [CrossRef] [PubMed]
- Azar, M.M.; Hage, C. Laboratory diagnostics for histoplasmosis. J. Clin. Microbiol. 2017, 55, 1612–1620. [CrossRef] [PubMed]
- Wheat, L.J.; Cloud, G.; Johnson, P.C.; Connolly, P.; Goldman, M.; Le Monte, A.; Fuller, D.E.; Davis, T.E.; Hafner, R.; AIDS Clinical Trials Group; et al. Clearance of fungal burden during treatment of disseminated histoplasmosis with liposomal amphotericin B versus itraconazole. Antimicrob. Agents Chemother. 2001, 45, 2354–2357. [CrossRef] [PubMed]
- Bahr, N.C.; Antinori, S.; Wheat, L.J.; Sarosi, G.A. Histoplasmosis infections worldwide: Thinking outside of the Ohio River Valley. Curr. Trop. Med. Rep. 2015, 2, 70–80. [CrossRef] [PubMed]
- Hage, C.A.; Ribes, J.A.; Wengenack, N.L.; Baddour, L.M.; Assi, M.; McKinsey, D.S.; Hammoud, K.; Alapat, D.; Babady, N.E.; Parker, M.; et al. A multicenter evaluation of tests for diagnosis of histoplasmosis. Clin. Infect. Dis. 2011, 53, 448–454. [CrossRef] [PubMed]
- Wheat, L.J.; Wass, J.; Norton, J.; Kohler, R.B.; French, M.L. Cavitary histoplasmosis occurring during two large urban outbreaks: Analysis of clinical, epidemiologic, roentgenographic, and laboratory features. Medicine 1984, 63, 201–209. [CrossRef] [PubMed]
- Wheat, J.; French, M.L.; Kohler, R.B.; Zimmerman, S.E.; Smith, W.R.; Norton, J.A.; Eitzen, H.E.; Smith, C.D.; Slama, T.G. The diagnostic laboratory tests for histoplasmosis: Analysis of experience in a large urban outbreak. Ann. Intern. Med. 1982, 97, 680–685. [CrossRef]
- Wheat, L.J.; Freifeld, A.G.; Kleiman, M.B.; Baddley, J.W.; McKinsey, D.S.; Loyd, J.E.; Kauffman, C.A. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2007, 45, 807–825. [CrossRef] [PubMed]
- Mazi, P.B.; Arnold, S.R.; Baddley, J.W.; Bahr, N.C.; Beekmann, S.E.; McCarty, T.P.; Polgreen, P.M.; Rauseo, A.M.; Spec, A. Management of histoplasmosis by infectious disease physicians. Open Forum Infect. Dis. 2022, 9, ofac313. [CrossRef] [PubMed]

